Tokyo Tech researchers have developed titanium dioxide nanotubes that outperform conventional catalysts in the synthesis of useful chemicals
Catalysts are a vital part of many industrial processes, speeding up chemical reactions to create useful products as efficiently as possible. One particular family, called Lewis acid catalysts, work by having the ability to accept pairs of electrons from the reactants. These catalysts, including aluminum chloride and boron trifluoride, can speed up the synthesis of important chemicals for manufacturing polystyrene, detergents, pharmaceuticals, cosmetics and even food additives.
Unfortunately, Lewis acid catalysts have their drawbacks. They tend to produce toxic waste, can be hard to separate from the final target product, and may corrode the manufacturing equipment. To this end, researchers have been investigating alternative, environmentally-friendly routes to Lewis acid catalysis.
Now, Masaaki Kitano and co-workers at Tokyo Tech’s Materials and Structures Laboratory have shown that tiny nanotubes made from titanium dioxide can outperform some of the best conventional catalysts1. More importantly, the nanotubes do not produce any toxic by-products, and can be reused many times.
“We have focused on titanium dioxide as a solid acid catalyst, because titanium dioxide contains Lewis acid sites and titanium is the second most abundant transition metal (ninth of all elements) in the Earth’s crust,” say the researchers, writing in the Journal of the American Chemical Society.
What’s more, titanate nanotubes can be produced by a simple, inexpensive hydrothermal process which requires no special chemicals. In recent years they have been used in sensors, lithium batteries and all sorts of catalysts including those activated by light or electricity. However, the applications in catalysis have generally made use of the nanotubes’ large surface area to support other chemicals for reactions. The Tokyo Tech team, working in collaboration with AIST in Tsukuba and Kanagawa Academy of Science and Technology, are the first to investigate the nanotubes’ potential for direct acid catalysis.
The researchers produced titanate nanotubes around 10 nanometers in diameter, containing extra protons and lots of pores along their lengths. They tested the catalytic performance of their protonated nanotubes for the reaction of toluene and benzylchloride, to make benzyltoluene, and compared it to other catalysts.
In tests at room-temperature, the older conventional catalysts showed negligible effects on the reaction. Some established Lewis acid catalysts called SO42+/ZrO2 and Hß gave a moderate enhancement to the reaction rate, but all were dramatically outperformed by the titanate nanotubes, which yielded 90% of the possible benzyltoluene in just 3 hours.
The researchers found no by-products after the reaction, and showed that the titanate nanotubes can be filtered, washed and reused without showing any loss in catalytic activity. This is a huge advantage over aluminum chloride which, although faster-acting, cannot be reused, and cannot exceed yields of 70% due to the formation of toxic by-products that get in the way of the reaction.
The nanotubes also outperformed other catalysts in enhancing the reaction between glucose and fructose to form HMF (5-hydroxymethylfurfural), an important molecule for the production of fuel, food and other materials from biomass.
On examining their nanotubes with spectroscopy, the researchers showed that, unlike ordinary titanium dioxide, the nanotubes contain not only Lewis acid sites but also ‘Brønsted acid’ sites, which can donate a proton. This makes the catalyst more efficient, as the researchers explain in their paper:
“Apparently, the alkylation by the Lewis acid sites at room temperature does not proceed without the Brønsted acid sites. Such synergy between Brønsted and Lewis acid sites has been reported to enhance the catalytic activity.”
Reference
- Kitano, M., Nakajima, K., Kondo, J.N., Hayashi, S. & Hara, M.
1. J. Am. Chem. Soc. 132, 6622-6623 (2010)
Materials and Structures Laboratory
Friedel-Crafts alkylation over various nanostructured titanate materialsa


a) HRTEM images of the protonated titanate nanotubes.
b) Enlarged HRTEM image of a).
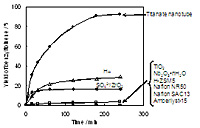
Time courses of benzyltoluene formation using various solid acid catalysts. Reaction conditions: catalyst (0.2 g), toluene (0.1 mol).
. Any information published on this site will be valid in relation to Science Tokyo.





