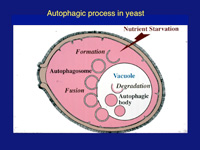
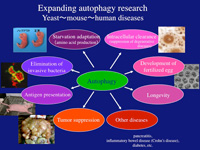
Autophagy—Greek for self-eating—is an efficient self-cleaning, biological revitalizing process used in our bodies. When cells face adverse conditions such as nutrient depletion, the autophagy process commences to enclose a portion of its own cellular constituents and delivers it to the lytic compartment (named lysosome in animals, vacuole in yeast and plants) for reuse.
Autophagy was first observed in the 1960s, when it was only known as a defense response against starvation. Recently, researchers discovered that autophagy also aids in combating harmful bacteria and viruses, protection against neurodegenerative diseases and cancer.
Yoshinori Ohsumi—a professor at Tokyo Institute of Technology—has played a pioneering role in the fundamental understanding of the autophagy process. Ohsumi began as a researcher in the University of Tokyo, then moved to the National Institute for Basic Biology and continues his work today at Tokyo Institute of Technology’s Frontier Research Center.
“When I began my research I was interested in how a yeast cell degrades its own constituents during its life cycle, and also how proteins were recycled in the vacuole,” says Ohsumi. Yeast, he points out, is one of the most suitable organisms for research in biology because in-spite of its relatively simple cellular structure, it contains almost everything found in our own cells. “So we can apply our understanding of it to animal and human cells,” he explains.
Ohsumi had the simple idea of using degradation-enzyme deficient mutant yeast cells and subjecting them to nitrogen starvation—a condition that would induce dramatic cell remodeling and spore formation. He expected this would result in extensive protein degradation.
“Using a standard light microscope, which is possible with yeast, I observed that this is exactly what happened,” says Ohsumi. Double membrane-bound spherical structures containing a portion of cytoplasm (the thick liquid holding the cell’s elements) form, and the outer membrane then fuses with the vacuole. This enables the cytoplasm to enter the vacuole, where it is processed, after which the digested products are returned to the cytoplasm for reuse. “This is a simple description of the autophagic process,” says Ohsumi.
Today it is known that harmful proteins and microorganisms invading the cell may also be dealt with in the same way. This knowledge is opening up the possibility of new treatments for cancer and infectious diseases, and other afflictions.
Ohsumi produced his findings more than two decades ago, a time when nothing was known about the genes responsible for autophagy. So he decided to employ genetic testing to screen mutant cells that cannot undergo autophagy: a procedure that would help him identify which genes are involved in the process.
To date Ohsumi’s group has identified 18 specific genes essential to autophagy in yeast. Significantly, these genes are well conserved from yeast to humans.
Once the autophagy genes and their encoding proteins are fully identified, researchers will be able to study what autophagy is accomplishing in our various organs. “So these findings can be considered the turning point in autophagic research,” says Ohsumi. “Now researchers can eliminate a particular gene essential for autophagy and see what happen to the cell’s functions.
When Ohsumi wrote his first paper on the subject in 1992, only 50 or 60 papers in total had been published. Today, there is intense interest in the field of autophagy and it’s estimated that some 2,000 papers on the subject were published in 2010.
The reason for the wide interest is not hard to understand. “In Nature an organism must maintain viability despite experiencing nutrient deficiency from time to time, therefore a recycling system of its own constituents is a fundamental requirement for life,” says Ohsumi.
Despite the tremendous interest in the field, there are still many unanswered questions. “Currently, we are working on finding out how the autophagosome membranes enclosing a portion of cytoplasm are formed and what makes them fuse with the vacuole,” says Ohsumi. Others are studying how a cell produces new membrane compartments and what determines their contents and size. Others still are looking to see how manipulating autophagy could control diseases.
“Many challenges remain,” says Ohsumi. “But within the next five years I believe we can overcome many of them.”
. Any information published on this site will be valid in relation to Science Tokyo.