Tailoring carbon chain lengths: conversion of two C5 fragments into C8 and C2 on a triruthenium cluster
A process that can yield hydrocarbons of desired chain length has been in continuous demand over the years. Recently, a method for converting lower alkanes into higher alkanes - referred to as alkane metathesis - has attracted considerable attention. Alkane metathesis involves three catalytic reactions: activation of the alkane C-H bond, olefin metathesis, and hydrogenation of the formed olefins. However, the method is hampered by cross-metathesis reactions of alkenes during the recombination process, which result in a molecular weight distribution in the product.
Now, Hiroharu Suzuki and colleagues at Tokyo Institute of Technology have shown that two C5 hydrocarbyl fragments can be selectively converted into C8 and C2 fragments using a triruthenium cluster as a model compound metal surface for the reaction. Thermolysis of the trinuclear µ3-pentylidyne-µ3-pentyne complex at 140 °C resulted in the quantitative formation of a µ3-octyne-µ3-ethlidyne complex.
Monitoring the reaction using 1H NMR spectroscopy revealed that the reaction proceeded by way of consecutive recombination of hydrocarbyl ligands on the triruthenium cluster concomitant with cleavage and reforming of the Ru-Ru bond (as shown in the Scheme). Thus the two C5 ligands coordinated on both faces of Ru3 plane were selectively converted into C2-alkylidyne and C8-alkyne fragments via a µ3-metallacyclobutenyl intermediate.
The results demonstrate the potential applicability of cluster compounds in synthetic reactions and shed light on the mechanism of reactions on metal surfaces.
Reference
- Authors: Atsushi Tahara, Mana Kajigaya, Makoto Moriya, Toshiro Takao, and Hiroharu Suzuki
- Title of original paper: Metathesis reaction of hydrocarbyl ligands across the triruthenium plane
- Journal, volume, pages and year: Angewandte Chemie International Edition 2010, 49, 5898-5901.
- Digital Object Identifier (DOI): 10.1002/anie.201002235
- Affiliations: Department of Applied Chemistry, Graduate School of Science and Engineering, Tokyo Institute of Technology
- Department website: http://www.apc.titech.ac.jp/index.html

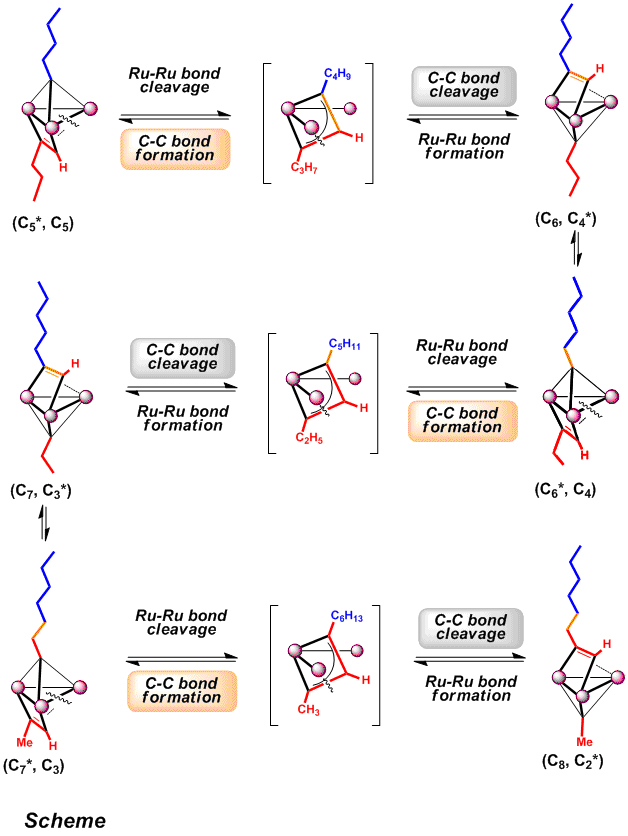
A plausible mechanism for the consecutive recombination of hydrocarbyl ligands on the Ru3 cluster.
. Any information published on this site will be valid in relation to Science Tokyo.



