Summary
Professor Takashi Nakamura, of the Department of Physics, Tokyo Institute of Technology, with RIKEN and international researchers, has clarified quantitatively three characteristic structure features of exotic nuclei - Neutron Halo(1), Disappearance of a Magic Number(2), and Strong Deformation. Measurements on the heavy, extremely neutron-rich Neon isotope(3) Neon 31 (31Ne) have provided a unified understanding of these phenomena.
The research involved a Tokyo Institute of Technology and RIKEN led international collaboration with the Seoul National University (Korea), University of Surrey (U.K.), Japan Atomic Energy Agency (JAEA), Laboratoire de Physique Corpusculaire de Caen (LPC-CAEN) (France), University of Tokyo Center for Nuclear Study (CNS), and Tokyo University of Science. The new results were published in the 7th April issue of "Physical Review Letters," (T. Nakamura et al., Physical Review Letters 112, 142501 (2014)), the foremost letters journal of the American Physical Society.
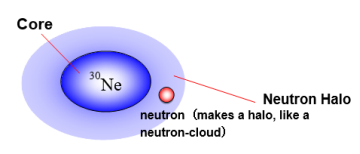
- Fig.1
- The diagram shows the observed features of the Neon 31 (31Ne) nucleus, with 10 protons and 21 neutrons. A dense 30Ne core nucleus is surrounded by a "neutron halo", where a single neutron forms an extended dilute cloud. It was shown that 31Ne is deformed, with a lemon-like shape, having a 3:2 (major: minor) axis ratio.
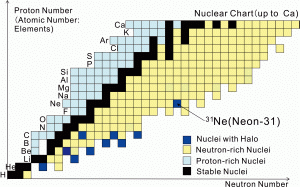
- Fig.2
- The nuclear (Segre) chart up to the Ca (calcium) isotopes. The horizontal axis represents the number of neutrons and the vertical axis the number of protons (the atomic number). The Neon 31 (31Ne) nucleus, with 10 protons and 21 neutrons, lies close to the boundary for the existence of bound neutron-rich nuclei (the neutron drip line).
Explanation of Technical Terms
(1) Neutron Halo
Unlike naturally occurring atomic nuclei, where all neutrons and protons are densely packed in a small volume, a neutron halo extends to larger distances, with a much lower density, surrounding a core nucleus with normal density; as shown schematically in Fig.1. Neutron halos are composed of one or two neutrons and are found in the most neutron-rich nuclei.
(2) Disappearance of a Magic Number
First explained by Nobel Laureates Maria Goeppert Mayer and J.Hans D.Jensen, conventional nuclei with Magic numbers (8, 20, 28, 50, 82, and 126) of neutrons or protons have extra stability compared to neighboring nuclei on the nuclear chart (Fig.2). Recent careful investigations, made possible by experiments using beams of rare isotopes (RI), have confirmed that the magic numbers 8 and 20 no longer exist in some neutron-rich nuclei, but can be replaced by new magic numbers; as recently evidenced in a RIKEN experiment on the heavy, neutron-rich calcium isotope 54Ca (Nature Nature(London) 502, 207 (2013).)
(3) Isotope
An atomic nucleus is composed of protons and neutrons, where the number of protons determines the associated chemical properties of the element and its atomic number. Different nuclei with the same number of protons are called isotopes, so 31Ne is an isotope of neon, all of which have 10 protons. Neon isotopes range from 17Ne to 34Ne of which only 20Ne, 21Ne, and 22Ne are naturally occurring stable isotopes found on earth. All other isotopes are unstable. 31Ne, with 21 neutrons, is an unstable neutron-rich isotope.
Reference
Authors: T. Nakamura et al
Title of original paper: Deformation-Driven p-Wave Halos at the Drip Line: Ne 31
Journal volume, pages and year: Phys. Rev. Lett. 112, 142501 - Published 7 April 2014
DOI: 10.1103/PhysRevLett.112.142501
. Any information published on this site will be valid in relation to Science Tokyo.




