Scientists at Tokyo Institute of Technology (Professor Masatomo Yashima and colleagues, Tokyo Tech) and Australian Nuclear Science and Technology Organisation (Dr. James R. Hester, ANSTO) have discovered a new structure family of pure oxide-ion conductors SrYbInO4. This new material is expected to lead to the development of solid oxide fuel cells, sensors, and oxygen separation membranes. This study would give a clue to solve energy and environmental problems in future.
Because some A2BO4-based materials such as (Pr,La)2(Ni,Cu,Ga)O4+δ exhibit high oxide-ion conductivity, scientists at Tokyo Tech have been exploring new structure families of ABCO4-based materials as BaRInO4, where R represents a rare earth element. Here, A, B, and C are cations located at different crystallographic sites, and A, B, and C in ABCO4 correspond to A, A, and B, respectively, in A2BO4.
Many researchers have investigated the optical, electrical, and magnetic properties of CaFe2O4-type materials, but the CaFe2O4-type pure oxide-ion conductors have not been reported yet. Therefore, Professor Masatomo Yashima and colleagues synthesized a new CaFe2O4-type material, strontium ytterbium indium oxide, SrYbInO4. They investigated its crystal structure from room temperature to 1273 K, its temperature and partial pressure dependence of electrical conductivity, and oxide-ion diffusion pathways. The occupancy factors are also carefully refined using not only conventional X-ray diffraction data but also time-of-flight (TOF) and angle-dispersive-type neutron and synchrotron X-ray diffraction data in order to obtain reliable results. They demonstrate a partial Yb/In occupational disorder in SrYbInO4 through careful analyses of occupancy factors.
Prof. Yashima and colleagues have chosen the chemical composition SrYbInO4, because it contains no transition-metal cation, which leads to less electronic conduction. Moreover, SrYbInO4 was expected to have the CaFe2O4-type structure in the structure field map shown in Fig. 1. Ionic radii of Sr2+ and (Yb3+, In3+) are larger than those of Ca2+ and Fe3+, respectively, thus the SrYbInO4 is expected to have a lower activation energy for oxide-ion conductivity compared with CaFe2O4.
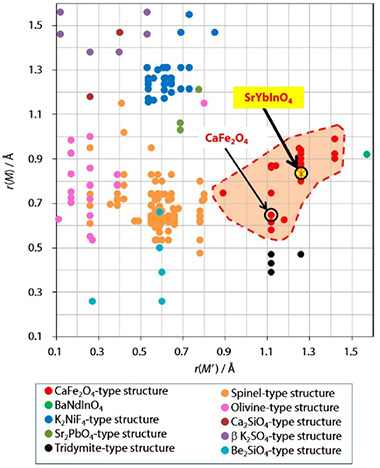
Figure 1. Structure Field Map of M2M′O4 Compounds.
The structure field map shows 209 different compositions of M2M′O4 compounds, with the red hatched area representing the CaFe2O4-type structure field. The newly synthesized compound SrYbInO4 is shown within the CaFe2O4-type structure field. ©American Chemical Society.
SrYbInO4 was synthesized by a solid-state reaction. SrYbInO4 was characterized through X-ray diffraction, chemical analysis, and thermogravimetric analysis. The band gap of SrYbInO4 was also estimated using UV-vis reflectance spectra, which suggested that SrYbInO4 is an electronic insulator. These results strongly suggested that SrYbInO4 was a pure oxide-ion conductor.
Using neutron and synchrotron X-ray diffraction data and Rietveld method, Prof. Yashima and colleagues showed that SrYbInO4 sample was a single orthorhombic phase with Yb/In occupational disordering at the B and C sites, and no vacancies at the cation and oxygen sites (Fig. 2). Bond valence sums and DFT-based structural optimization indicated the validity of the refined crystal structure of SrYbInO4. Therefore, the new material SrYbInO4 is the first example of pure oxide-ion conductors with a CaFe2O4-type structure.
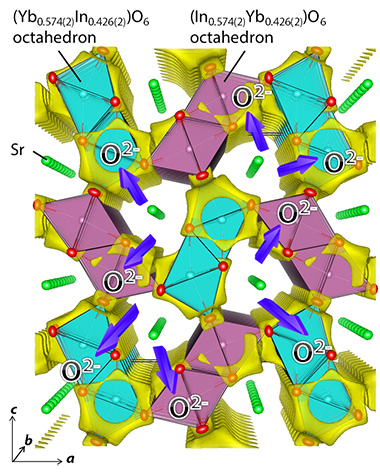
Figure 2. Refined Crystal Structure and Bond-Valence-Based Energy (BVE) Landscape of a test Oxide Ion of SrYbInO4.
Yellow isosurface of the BVE at 0.8 eV suggests that the oxide ion migrates in the b direction along the edge/surface of light blue/purple octahedra BO6 [B = Yb0.574(2)In0.426(2)] and purple octahedra CO6 [C = In0.574(2)Yb0.426(2)] where the number in the parenthesis is the estimated standard deviation. ©American Chemical Society.
Further the temperature dependence of oxide-ion conductivity showed lower activation energy of SrYbInO4 (1.76 eV) than that of CaFe2O4 (3.3 eV), which was supported also by the bond valence-based energy calculations. The lower activation energy is attributable to the larger bottleneck size for oxide-ion migration due to the larger ionic radii of Sr2+ and (Yb3+, In3+) than those of Ca2+ and Fe3+, respectively.
Prof. Yashima and colleagues claimed that the oxide ion conductivity of SrYbInO4 could be improved by doping, changing the degree of cation ordering and disordering, and using larger A, B, and C cations in the ABCO4-structure, which leads to further lowering the activation energy and higher oxide-ion conductivity. The findings of this study may open new pathways in the development of ABCO4-based ion conductors.
Reference
Authors : |
Ayaka Fujimoto1, Masatomo Yashima1,*, Kotaro Fujii1, James R. Hester2 |
Title of original paper : |
New Oxide-Ion Conductor SrYbInO4 with Partially Cation-Disordered CaFe2O4-Type Structure |
Journal : |
The Journal of Physical Chemistry C |
DOI : |
|
Affiliations : |
1Department of Chemistry and Materials Science, Graduate School of Science and Engineering and Department of Chemistry, School of Science, Tokyo Institute of Technology, Japan
2Australian Centre for Neutron Scattering, Australian Nuclear Science and Technology Organisation (ANSTO), Australia
|
. Any information published on this site will be valid in relation to Science Tokyo.





