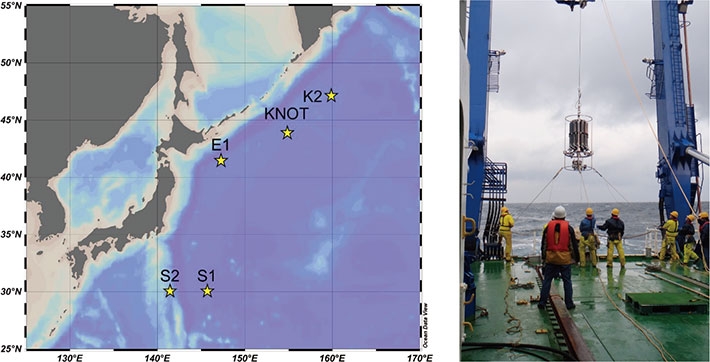
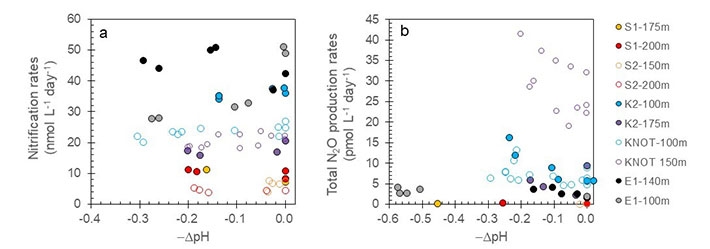
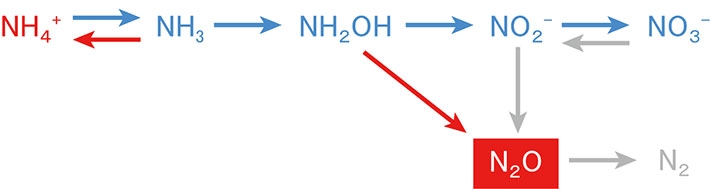
1Department of Environmental Chemistry and Engineering, Tokyo Institute of Technology, Nagatsuta 4259, Midori-ku, Yokohama, 226-8503 Kanagawa, Japan
2Institute of Environmental Engineering, Ecole Polytechnique Fédérale de Lausanne - EPFL, Station 2, CH-1015 Lausanne, Switzerland
3Research Institute for Marine Resources Utilization, Japan Agency of Marine Earth Science and Technology, 2-15 Natsushima-cho, Yokosuka-city 237-0061, Japan
4Institute for Extra-cutting-edge Science and Technology Avant-garde Research (X-star), Japan Agency of Marine Earth Science and Technology, 2-15 Natsushima-cho, Yokosuka-city 237-0061, Japan
5School of Materials and Chemical Technology, Tokyo Institute of Technology, Nagatsuta 4259, Midori-ku, Yokohama, 226-8503 Kanagawa, Japan
6Research Institute for Global Change (RIGC), Japan Agency of Marine Earth Science and Technology,2-15 Natsushima-cho, Yokosuka-city 237-0061, Japan
7Atmosphere and Ocean Research Institute, The University of Tokyo, 5-1-5, Kashiwanoha, Kashiwa-shi, Chiba 277-8564 Japan
8Earth-Life Science Institute, Tokyo Institute of Technology, Meguro, 152-8551 Tokyo, Japan
. Any information published on this site will be valid in relation to Science Tokyo.