Chemists at Tokyo Tech propose an innovative way to achieve carbon capture using a rhenium-based electrocatalytic system that is capable of reducing low-concentration CO2 (even 1%) with high selectivity and durability, which is a new potential technology to enable direct utilization of CO2 in exhaust gases from heavy industries.

Figure 1. Photograph of the experimental setup for bulk electrolysis
The CO2 reduction reaction takes place in the cathodic chamber shown on the right.
Scientists are closer to finding effective ways to reduce CO2 levels — a vital part of responding to climate change and energy efficiency challenges.
A study led by Osamu Ishitani of the Department of Chemistry, Tokyo Tech now demonstrates the advantages of applying electrocatalysis1
to capture low-concentration CO2.
In their study published in Chemical Science, Ishitani and colleagues including Hiromu Kumagai and Tetsuya Nishikawa drew on decades of work on honing the capabilities of a rhenium-based catalyst, and demonstrated its ability to reduce low-concentration CO2 in the presence of a chemical called triethanolamine (TEOA).
Compared to many previous studies that have focused on reducing pure CO2, few have explored how to improve direct capture of low-concentration CO2 — a topic that warrants further investigation, considering that plants harness low concentrations of CO2 (about 400ppm, that is 0.04% of the atmosphere) and exhaust gases from heavy industries typically contain low levels of CO2 (around 3–13%).
By avoiding the need for additional energy-consuming condensation processes, their strategy, if scaled up, could provide a more viable, environmentally friendly solution to CO2 capture in many settings.
In a series of experiments (see Figure 1) to assess electrocatalytic activity, the researchers found that at a CO2 concentration of 1%, the rhenium-based catalyst showed very high selectivity (94%) towards carbon monoxide (CO) formation (see Figure 2).
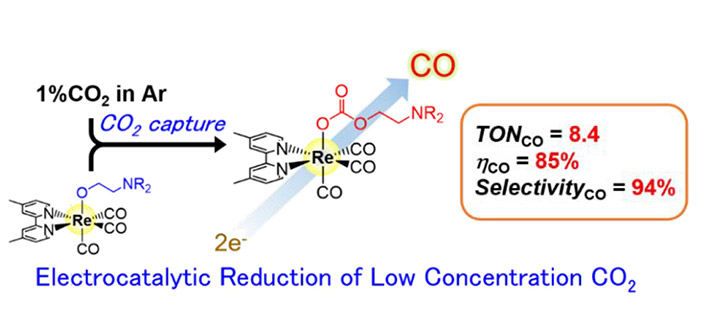
Figure 2. Representation of the electrocatalytic reduction system
Electrocatalytic reduction of low-concentration CO2 was achieved using a rhenium-based complex with high CO2-capturing ability.
A likely reason behind the high performance, the researchers say, is the efficient insertion of CO2 into the rhenium–oxygen bond.
The researchers aim to continue systematically investigating promising strategies to help reduce real-world CO2 levels.
The present study was supported by a CREST (Core Research for Evolutional Science and Technology) program on Molecular Technology backed by the Japan Science and Technology Agency (JST).
The paper has been selected as part of the 2018 ChemSci Pick of the Week Collection and a news story are posted on News & events
and a news story are posted on News & events of Royal Society of Chemistry.
of Royal Society of Chemistry.
1 Electrocatalysis
A process that harnesses electrochemical reactions to accelerate a particular reaction of interest.
Reference
Authors : |
Hiromu Kumagai, Tetsuya Nishikawa, Hiroki Koizumi, Taiki Yatsu, Go Sahara, Yasuomi Yamazaki*, Yusuke Tamaki and Osamu Ishitani |
Title of original paper : |
Electrocatalytic Reduction of Low Concentration CO2. |
Journal : |
Chemical Science (2018) |
DOI : |
|
Affiliations : |
Department of Chemistry, School of Science, Tokyo Institute of Technology
*Present address: Faculty of Science and Technology, Department of Materials and Life Science, Seikei University
|
. Any information published on this site will be valid in relation to Science Tokyo.





